Introduction
13 April 2008: Miscellaneous minor edits.

| At a Glance | |
|---|---|
| Product | MozyHome Online Backup |
| Summary | Simple yet customizable online backup at an affordable price |
| Pros | • Good balance of features and simplicity • Inexpensive • Unlimited storage and transfers |
| Cons | • Slow uploads to Mozy no matter how fast your upstream is • Backups run as batch operations, not continuously • Restores take longer than expected and files are hard to identify |
Data backup is one of the most important aspects of IT, yet so few people think about it until it’s too late. To solve this problem, Mozy created an online backup package as automatic and unobtrusive as possible. As with all consumer solutions, it has its strengths and weaknesses. Companies like Mozy have to balance easy-of-use with configurability, and decide what their program does best, all while trying to make money.
While there are many backup options available, rarely have there been any as simple to use as MozyHome. The MozyHome service is designed to automatically back up data files from your computer to Mozy’s online servers. The only potential problem with Mozy’s extreme ease-of-use is that it is only intended to back up data files, not entire systems. It is not meant to be a comprehensive backup solution for every situation, merely a way to recover home user-type files such as Word documents and mp3s.
Setup
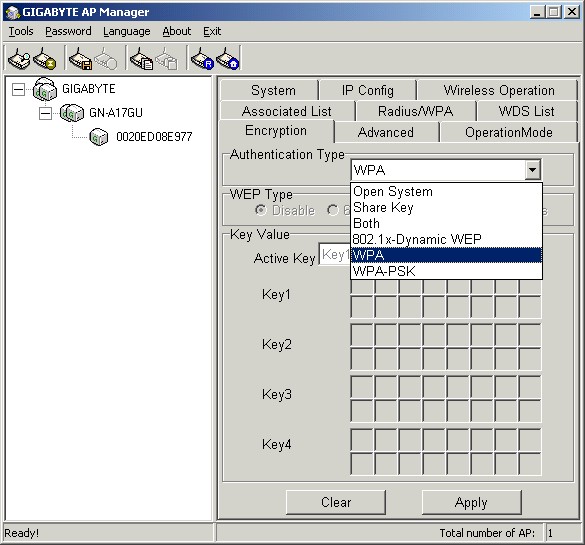
Because Mozy is intended to be easy to use, setup can be quite simple. All you really have to do is select whether you want to use Mozy’s encryption key or your own encryption key (Figure 1), select the data to back up, and select the performance options you want. After that, Mozy can take care of the rest, or you can configure advanced options.
Figure 1: Selecting the encryption key
Although Mozy recommends using their key, so that they can recover your files for you, security-conscious users might realize that this means they can access your files. Although their terms of use and privacy policy say that they won’t, it is possible that they can; and, of course they would have to access them if required by law enforcement officials.
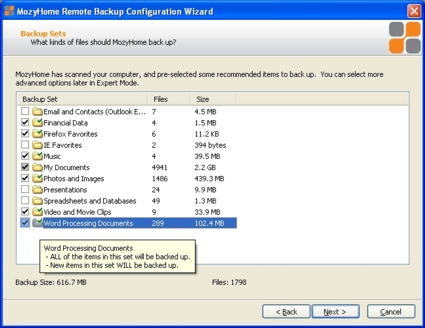
After the encryption key option is set, Mozy will ask you which backup sets you want to back up (Figure 2). There are more backup sets available depending on the types of data Mozy finds on your computer, and you can create custom backup sets later.
Figure 2: Selecting backup sets to include
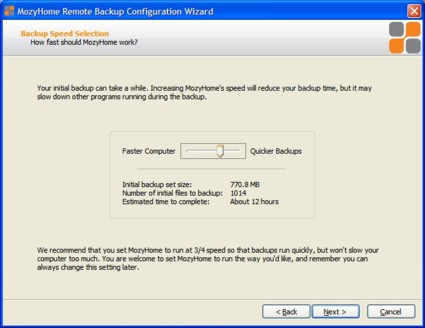
Once your backup is configured, the only thing left to do is set the backup performance option (Figure 3). The effects of this option will be discussed in the Performance section.
Figure 3: Choosing backup performance options during setup
Features
As mentioned before, Mozy has some nice features and some trade-offs. One of its nice features is the fact that it installs a virtual drive icon (Figure 4) that shows you all of the files you have backed up on Mozy’s servers. A benefit of this feature is the ability to search for deleted or corrupted files using the Search function in Windows.
Although you can’t do anything with the search results—such as restoring the file from the results list or opening the containing folder—you can still find files that were accidentally deleted so that you can restore them.
Figure 4: Mozy Virtual Drive icon in Windows Explorer
Figure 5 shows the entire list of configuration options.
Figure 5: Configuration options for MozyHome
The Bandwidth Throttle and Backup Speed options provide an easy way to ensure that you won’t even notice Mozy operating. Mozy does not seem to put much of a strain on system resources even in the most aggressive settings (see Performance section), but it’s still handy to have sliders that let you adjust the impact on performace if it becomes bothersome.
The only other configuration settings Mozy has are for scheduling backups. The settings, as seen in Figure 6, are fairly simplistic. It’s important to note that there’s no way to run continuous backups whenever files change. Mozy will back up your data, at most, 12 times per day, and then only if the computer has been idle for at least a minute.
The only way to force Mozy to perform a backup when the computer is not idle is to initiate a manual backup from the Mozy agent or to schedule a daily or weekly backup. It would be nice to see an option to backup files whenever they change, or at least to schedule hourly backups, although Mozy probably doesn’t want that much strain on their servers.
Figure 6: Backup scheduling options
Disappointments
Now for the disappointing aspects of Mozy, at least from an advanced user’s perspective.
MozyHome does not support backing up network drives or removable storage. The network drive limitation makes sense, since MozyHome is billed per computer. If you could back up network drives, there would be no reason to pay for multiple computers. Mozy’s lack of support for external drives (unless the computer recognizes them as fixed storage) makes sense when you consider that most people probably only use external drives as temporary or backup storage in any case.
One confusing aspect of Mozy is that the backup set information as displayed is not particularly helpful. With its focus on simplicity, Mozy organizes files in "sets", which are organized by file type. Referring back to Figure 2, the data displayed beside each backup set refers to the total possible size of the set. But if you select only some of the files in a set to be backed up, the Backup Set Size number doesn’t change.
Let’s say you’re a photographer who wants to back up some critical pictures, but also wants to throw in some other files that aren’t as critical, but that you would rather not lose. You’ll obviously include the pictures no matter how much space they take up, but you notice that the total backup size is around 9 GB, which will take a long time in the initial backup stage. You want to trim that down, but how can you decide which files or backup sets to remove if you don’t know how much space they’re actually using in the final size count? The only apparent way to figure out actual usage for a backup set that is only partially selected is to navigate through the file structure and add up the individual file sizes.
There is also no obvious way to force a new full backup after your initial one. Given the small size of the "pipe" between your computer(s) and Mozy’s servers, this isn’t something you would want to do very often. But if you made major changes, say a full system restore, and wanted to start from scratch, you would probably take the hit of the time it takes to do an initial full backup.
Finally, while Mozy has no limit on the number of different versions it keeps of the same file, it discards any versions over 30 days old, with no ability for user intervention or control.
Restore Options
There are a few ways to restore your files if you still have access to the computer on which Mozy is installed. The first is to browse through the virtual drive and click on the file you want to restore. Once you click on the file, you’re presented with a list of the file’s versions, although there’s no way to tell when the version was created or modified. Once you pick a version, you can either have it overwrite the file on your computer or have it renamed for you and placed in the same directory as the original; alternatively, you can have it restored to another location and select a name for it. It’s not the most flexible system, but it does work.
Another way to accomplish the same thing is to right-click inside a folder containing a file you want to restore and select “Restore Files in Folder” from the shortcut menu. This way is easier than browsing through the directory structure of the virtual drive if you already have the necessary folder open.
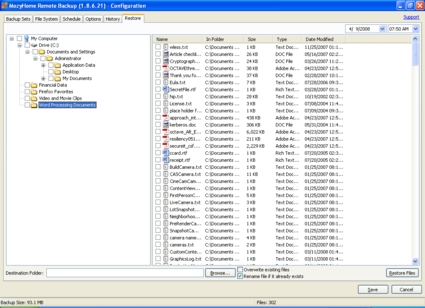
The final restoration method is through the Mozy agent (Figure 7). Restoring through the Mozy agent is similar to restoring via the Web Restore feature (covered below). You can browse the backup by file structure—fairly cumbersome—or by backup set—preferred if you know what type of files you want to restore.
Figure 7: Starting a restore operation via the Mozy agent
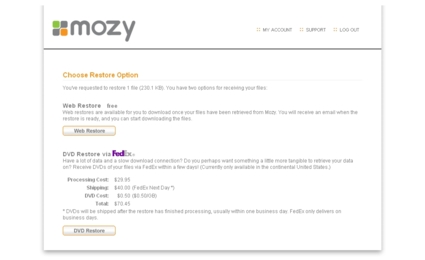
If you are not at your computer, you can still restore files from it via two Mozy options available in your account on mozy.com. Here, you can pick the backup date (up to 30 days previously), then select the files you want to restore. You are then presented with two options for restoration (Figure 8).
Figure 8: Online restore options on mozy.com
The Web Restore option builds your restore request, places the files in a.zip file and emails a notification that your request has been processed. In a simple test for the one 230 K file in Figure 8, the file was ready within about four minutes in the Restores section of the mozy.com Account page. For some reason, however, the email notification took around 32 minutes to arrive. For larger restores, it can obviously take quite a long time to prepare the.zip.
The other online restore option is DVD Restore, which allows you to select files to be placed on a DVD or set of DVDs and shipped to you. This option is not smart or cost-effective for anything less than critical data, because as you can see from Figure 8, it costs a lot. Otherwise, you can just download or restore it using one of the other methods Note that if you chose to use your own encryption key, you’ll need to download the decrypt tool on your Mozy account download page and enter your key to recover your files using either of the online methods.
Performance
Measuring the performance of an online backup tool is a bit problematic, especially in Mozy’s case. For one thing, it depends partly on your geographical position in relation to their data center (in Salt Lake City, Utah). For another, since Mozy has customizable settings that allow you to control the amount of system resources and bandwidth it uses, performance changes with different settings.
That being said, however, even on an older system, performance was good. Mozy used around 17 MB of memory when actively running a backup, and CPU usage spiked to about 45% when set to the most aggressive setting; however, there was no noticeable slowdown. The most aggressive setting did, however, slow down a six-year-old laptop, which is expected. As for bandwidth, Mozy reports that most people average around 200–500 Kbps to their servers.
As you can see in Figure 9, backing up about 770 MB is expected to take over 12 hours. However, in actual practice, this backup only took around six-and-a-half hours. The DSL connection used has an upstream of 512 Kbps. Mozy states that the initial backup could take days or weeks, depending on the connection speed and amount of data.
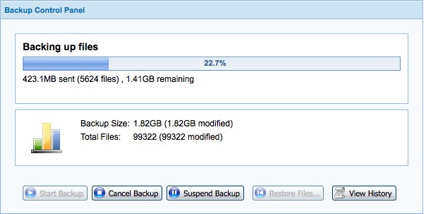
Figure 9: A backup in progress
Restoring, on the other hand, took longer than expected. A restore of the entire backup (779 MB) took over two hours and 20 minutes on a 3 Mbps connection: about the amount of time it would take a normal file transfer on a 768 Kbps connection. Most users probably don’t need to restore that much data at one time, but if they do, they should be prepared to wait.
Pricing and Conclusion
The price for MozyHome ranges from free for storing up to 2 GB from up to two computers, to $4.95/month (per computer) for unlimited transfers/restores/data. Signing up for a year will cost $54.45, but save you $4.95, while signing up for two years will cost $103.95, but save you $14.85.
Although the storage space is "unlimited", Mozy’s website notes that after storing about 200,000 files, performance will start to degrade to the point where it is unacceptable. Most home users shouldn’t have much of a problem with the space offered, unless they try to use it for full backups or commercial purposes.
Mozy offers Pro and Enterprise versions of the software for business users. Among their other features, both add support for Windows Server 2000 and 2003 to MozyHome’s support for Windows 2000, XP and Vista (and Mac OS X 10.4). They also add the ability to back up SQL, Exchange and file servers as well as networked shares and mapped drives. This flexibility comes at a price, however, namely $3.95 + $0.50/GB per month for desktop Pro licenses and $6.95 + $1.75/GB per month for servers.
An important point to remember is that although Mozy includes unlimited data storage for your backups, this does not mean you can store unlimited data. The only data stored on mozy.com is that which the MozyHome agent backs up. There is no file transfer utility, and deleted files are only stored for 30 days. Neither of these things mean Mozy is a bad deal; however, it does mean that it is not a complete replacement for normal backups. The flip side of the equation is that you don’t pay per GB for storage or transfers.
Although MozyHome is not a perfect solution that will satisfy every user, it does provide a compelling and easy backup method for normal home users who otherwise might not have a copy of their data if they lost it. With low monthly prices, easy-of-use, unlimited storage, and good features, Mozy offers a product that’s hard to ignore.